Metabolic dysfunction-associated steatotic liver disease (MASLD) now affects more than one-third of the global adult population and has become an increasingly important driver of cirrhosis, hepatocellular carcinoma, liver transplantation, and liver-related mortality. The newly accepted global consensus recommendations in Clinical Gastroenterology and Hepatology (2026) provide a practical roadmap to help clinicians move from passive disease recognition to proactive, risk-stratified management.
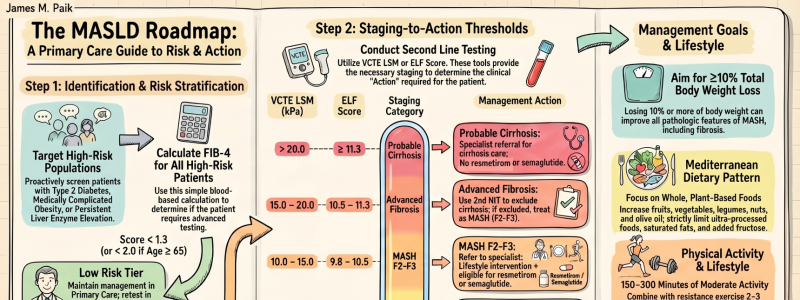
Central to this paradigm shift is a fundamental principle: fibrosis risk, rather than steatosis alone, should guide clinical decision-making. Patients with type 2 diabetes, medically complicated obesity, persistent aminotransferase elevation, or multiple cardiometabolic risk factors warrant active risk stratification using first-line noninvasive testing with FIB-4. This approach enables primary care clinicians to rapidly distinguish low-risk patients suitable for routine management from those requiring further assessment and potential specialist referral.
The algorithm employs a two-tiered stratification strategy. Second-line noninvasive tests, vibration-controlled transient elastography (VCTE) or the enhanced liver fibrosis (ELF) score, further define disease severity and guide actionable decisions. These tools identify patients with low-risk disease, those with at-risk MASLD and clinically significant fibrosis (F2–F3) where intervention is most impactful, and those with probable cirrhosis who require specialist management. Patients with F2–F3 fibrosis without cirrhosis may be considered for MASH-approved regimens in addition to lifestyle modification. Those with cirrhosis, while not currently candidates for approved MASH pharmacotherapies, should be referred for specialist management, whereas patients with earlier-stage disease (F1) can generally remain under primary care management. Critically, this framework transforms noninvasive tests from diagnostic tools into treatment-enabling instruments.
For the first time, the consensus also provides clear threshold-based recommendations for both VCTE and ELF in risk stratification and treatment selection. It further integrates recently approved pharmacotherapies, resmetirom and semaglutide, for appropriate noncirrhotic patients with moderate-to-advanced fibrosis (F2–F3), while emphasizing foundational lifestyle interventions: weight reduction, Mediterranean-style dietary optimization, alcohol minimization, reduction of ultra-processed foods and sugar-sweetened beverages, and regular physical activity. Longitudinal monitoring with repeat noninvasive testing during follow-up or active treatment allows objective assessment of treatment response and refinement of management over time.
The future of MASLD care is decisively noninvasive, risk-stratified, scalable, and actionable, combining evidence-based pharmacotherapy with optimized lifestyle modification and comprehensive cardiometabolic risk management for eligible patients.
Read the full article: https://www.cghjournal.org/article/S1542-3565(26)00241-7/fulltext